Research Moving tuberculosis vaccines from theory to practice

Professor Tom Scriba has co-authored a paper titled: "Moving tuberculosis vaccines from theory to practice" appearing in the Nature Reviews Immunology Journal.
Tuberculosis (TB) vaccine research has reached a unique point in time. Breakthrough findings in both the basic immunology of Mycobacterium tuberculosis infection and the clinical development of TB vaccines suggest, for the first time since the discovery of the Mycobacterium bovis bacillus Calmette–Guérin (BCG) vaccine more than a century ago, that a novel, efficacious TB vaccine is imminent. Here, we review recent data in the light of our current understanding of the immunology of TB infection and discuss the identification of biomarkers for vaccine efficacy and the next steps in the quest for an efficacious vaccine that can control the global TB epidemic.
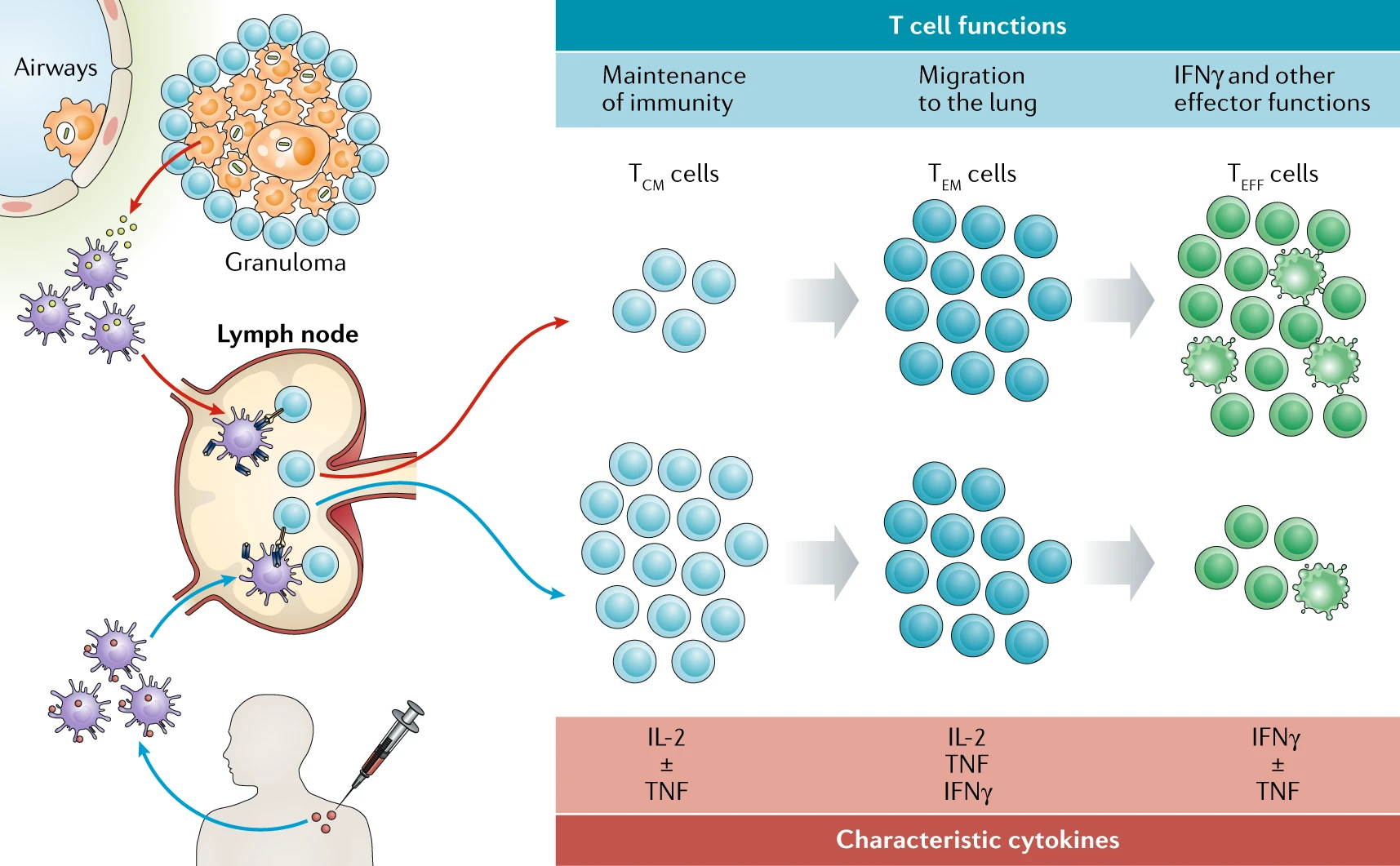
After Mycobacterium tuberculosis (Mtb) infection of alveolar macrophages (red arrows), Mtb is transported to lung-draining lymph nodes by infected dendritic cells, initiating T cell priming or triggering the activation of pre-existing memory T cells, which appears to preferentially drive T cell differentiation towards late-stage T effector memory (TEM) cell and T effector (TEFF) cell responses. Ongoing antigen expression is likely a driver of this T cell differentiation process, which favours primarily IFNγ -expressing and/or TNF-expressing T cells and little IL-2 expression. Vaccine administration in the skin or muscle promotes antigen uptake by dendritic cells, which traffic to draining lymph nodes to prime or activate T cells (blue arrows). In the case of subunit protein-adjuvant vaccines, the resulting T cell responses appear to be dominated by less differentiated T central memory (TCM) cell responses; these cells primarily express IL-2 and/or TNF. Achieving long-lived protective immunity by vaccination may require the establishment of a careful balance between TCM cell and TEM cell responses, such that a durable pool of memory cells resides in secondary lymphoid tissues while an appropriate tissue-resident population with rapid effector function is maintained in peripheral airway tissues.
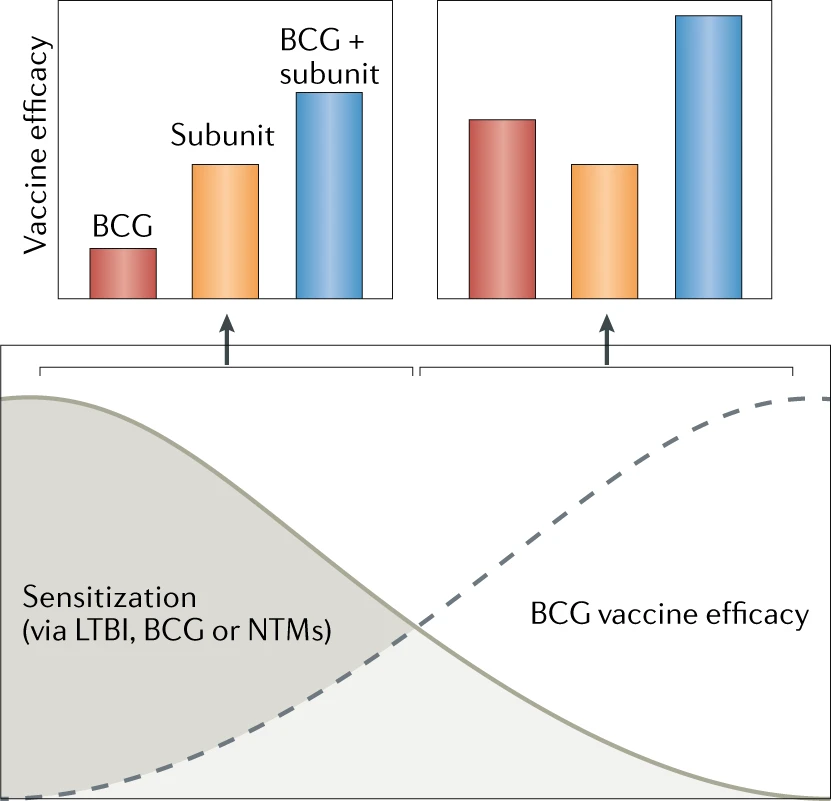
The hypothesized interaction between the magnitude of immune sensitization and vaccine efficacy by Mycobacterium bovis bacillus Calmette–Guérin (BCG) vaccination (red), adjuvanted protein subunit vaccines (orange) or a BCG-prime, subunit-boost strategy (blue) is shown. According to this model, BCG, and other live whole mycobacterial vaccines, are not efficacious in individuals with substantial prior immunological sensitization owing to latent tuberculosis infection (LTBI), recent BCG vaccination or exposure to atypical non-tuberculous mycobacteria (NTMs) from the environment. Subunit vaccination, by comparison, would be efficacious in such a pre-sensitized population, as would a BCG-revaccination, subunit-boost strategy. In persons with low or no mycobacterial sensitization, the efficacy of BCG vaccination is significantly increased, and the efficacy of subunit vaccination will be largely independent of the levels of sensitization, but the BCG-revaccination, subunit-boost strategy may provide synergistic effects that result in enhanced vaccine efficacy.
The large number of different vaccine candidates and their advanced stages in clinical development denote a unique and exciting phase in TB vaccine research. There are also a large number of novel vaccine candidates in preclinical development, including more recently developed vaccine formats such as DNA vaccines, new adjuvants and delivery systems and combination vaccines. It is important that the most promising of these new candidates are advanced to efficacy studies in animals and clinical testing to augment the pipeline of TB vaccine candidates and concepts.
However, a notable limitation of the current clinical development landscape is a lack of inter-trial harmonization or standardization, which precludes a direct comparison of the immunological outcomes of different TB vaccine candidates. A recent analysis attempted to tackle this problem by comparing antigen-specific CD4+ and CD8+ T cell responses induced by BCG and six of the novel TB vaccine candidates, including MVA85A, AERAS-402, H1:IC31, H56:IC31, M72/AS01E and ID93 + GLA-SE. The investigators retrieved published data on antigen-specific T cell responses from clinical trials completed in adolescents or adults at a single trial site in South Africa82. The results show that the magnitude of vaccine-induced TH1 cell-polarized CD4+ T cell responses measured several months after vaccination was the T cell response feature that diverged the most between the different candidates. Unlike the response magnitude, co-expression profiles of IFNγ, TNF and IL-2 by CD4+ T cells suggested a relative lack of functional diversity in responses induced by the different vaccine candidates (see Table 1). Interestingly, the analyses suggested that M72/AS01E induced the highest antigen-specific memory CD4+ cell response among the candidates. Unfortunately, the study did not include results from whole cell or live vaccine candidates, which are known to induce a more diverse and broader repertoire of immune responses.
Overall, the recent positive clinical trial data referred to above represent a very important milestone in international efforts to develop a novel efficacious TB vaccine. These successes illustrate that TB vaccine research is on the right track and will be able to deliver a much-needed improved vaccine strategy that is so critical for controlling the global TB epidemic. It is critical that the field moves forward with urgency towards phase III licensure trials so that an impact on the epidemic can be achieved swiftly. These findings therefore signal the end of the ‘post-MVA85A period’ of fundamental doubts about both the usefulness of the TB vaccine research strategy and the TB animal models in vaccine discovery83. The M72/ASO1E vaccine contains only two antigens, and the next generation of vaccines may be improved by adding more antigens to increase immune coverage and avoid the risk of escape. The efficacy signal observed with M72/ASO1E will likely also establish the M72/ASO1E-induced protection as a minimum benchmark in preclinical animal models. As discussed above, many subunit vaccine candidates appear to induce a response that is typically characterized by early differentiated CD4+ TCM and TEM cells, whereas it seems that both viral and live mycobacterial vectors promote a more differentiated CD4+ TEFF cell response27,52,54,82,84,85. It will therefore be important to agree on a standard set of parameters that would allow an accurate comparison between studies and vaccines to determine whether this is a reproducible pattern in clinical trials. Recent results of preclinical studies using a recombinant human cytomegalovirus encoding several Mtb T cell antigens have shown impressive protection in an NHP model, where prophylactic vaccination prevented infection in one-third of experimentally infected rhesus macaques86. Because cytomegalovirus vectored vaccines establish a persistent lifelong infection and induce a high level of antigen-specific CD4+ and CD8+ T cell responses, these findings suggest that, in addition to T cell differentiation as discussed above, the sheer size of the pool of Mtb-specific CD4+ T cells has an impact on protection. This is also supported by the M72/ASO1E trial results, as this vaccine promotes a high frequency of antigen-specific T cells. However, more is not always better, and there is a risk of less protection and insufficient immune memory or even of inflicting immunopathology with vaccines that induce very strong T cell responses, particularly when used in the postexposure setting in individuals with LTBI. With an efficacy signal in young adolescents and/or adults from both BCG revaccination and subunit vaccine studies, it is intriguing to speculate whether the combination of both, administered sequentially or simultaneously87, may pave the way for a vaccination strategy that protects both uninfected and infected people while providing the possibility of a synergistic effect for inducing more diverse and broader-ranging immune responses. If an additive effect can be demonstrated, the combination of BCG and subunit vaccines may represent a new strategy that elevates the efficacy signal into a range that triggers clinical implementation.
