New modelling study shows impact of late stage TB vaccines depends on their ability to prevent infectious, asymptomatic TB

New modelling study shows impact of late stage TB vaccines depends on their ability to prevent infectious, asymptomatic TB
New research—co-authored by SATVI’s Professor Mark Hatherill—has found that the real-world impact of late-stage tuberculosis (TB) vaccines may depend heavily on how well they protect against infectious, asymptomatic TB.
Why Asymptomatic TB Matters
Asymptomatic TB occurs when a person has active TB disease, but does not display classic symptoms such as a persistent cough, weight loss, night sweats, or loss of appetite. People with asymptomatic TB would usually not present to clinics for TB investigation. This makes diagnosis difficult and some cases eventually progress to symptomatic TB.
A recent study by Mendelsohn (2025) found that five out of ten people with active TB have no symptoms. Previous work by Moyo et al. has shown that up to half of all people with active TB may be asymptomatic, meaning a large proportion of transmission could be coming from individuals who do not yet feel ill. This “silent reservoir” is a key challenge for global TB control efforts—and a critical consideration for vaccine development.
Understanding the Study
Two promising TB vaccine candidates, M72/AS01E and MTBVAC, are currently in Phase 3 trials. While both have shown encouraging results so far, their long-term population impact remains uncertain. This study set out to better understand that potential by examining how vaccine impact might vary depending on which stages of TB disease they prevent.
Researchers used a mathematical model to explore three possible ways a vaccine might interrupt the progression of TB:
- Preventing only infectious, symptomatic TB disease
- Preventing any infectious TB disease (symptomatic or asymptomatic)
- Preventing all forms of TB disease
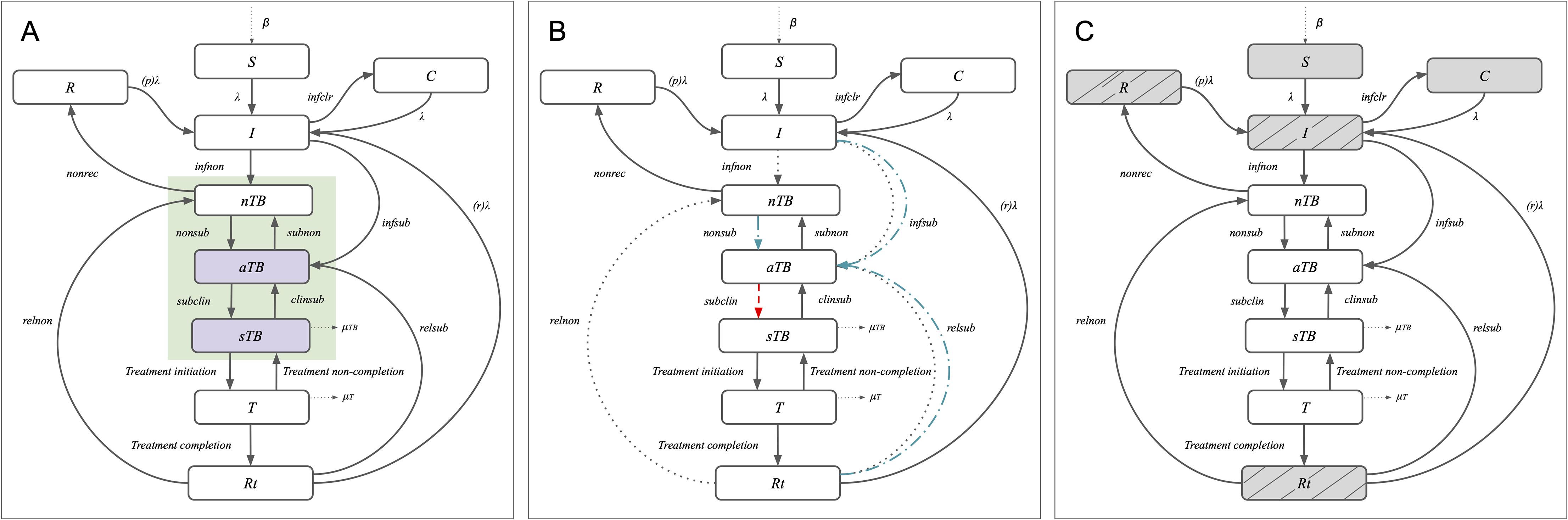
Source: Tanvir et al, Plos One, 2026
Over the long term, vaccines that prevented any infectious TB—or all TB disease—had a much greater impact on reducing the total number of symptomatic TB cases and TB deaths than those preventing only infectious, symptomatic TB. This is likely because asymptomatic TB cases that are prevented do not have the opportunity to transmit new infections, or progress to symptomatic disease with associated morbidity and mortality.
Key Takeaways
The study highlights that:
- The population-level benefits of new TB vaccines may depend on how effectively they prevent infectious asymptomatic disease, not only symptomatic disease.
- Vaccine trials should include measures of impact on asymptomatic TB to better predict real-world effectiveness and understand the full value of new vaccines.
- More data is needed to refine models, particularly about the natural history and infectiousness of asymptomatic TB.
As Phase 3 trials for M72/AS01E and MTBVAC continue, these insights underline the importance of designing vaccines—and new studies—that address all stages of TB disease, including the often-overlooked asymptomatic phase.
